Seznamy 93+ Structure Of Atom Of Fluorine Zdarma
Seznamy 93+ Structure Of Atom Of Fluorine Zdarma. It is part of the second period and seventeenth group. · cross section (thermal neutron capture)a/ barns: · electrons per energy level:
Nejlepší Fluorine Atom Images Stock Photos Vectors Shutterstock
It is part of the second period and seventeenth group. 9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns:17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.
9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has nine protons, nine electrons, and ten neutrons. Nine electrons (white) occupy available electron shells (rings). 9), the most common isotope of the element fluorine. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · electrons per energy level: · cross section (thermal neutron capture)a/ barns:

· cross section (thermal neutron capture)a/ barns: · cross section (thermal neutron capture)a/ barns: The nucleus consists of 9 protons (red) and 10 neutrons (orange). It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons. I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). · cross section (thermal neutron capture)a/ barns:

The nucleus consists of 9 protons (red) and 10 neutrons (orange)... · cross section (thermal neutron capture)a/ barns: It has nine protons, nine electrons, and ten neutrons. · electrons per energy level: I show you where fluorine is on the periodic table and how to determine. It is part of the second period and seventeenth group.

Nine electrons (white) occupy available electron shells (rings). It has nine protons, nine electrons, and ten neutrons. · electrons per energy level: 9), the most common isotope of the element fluorine. It is part of the second period and seventeenth group. I show you where fluorine is on the periodic table and how to determine. · cross section (thermal neutron capture)a/ barns:. It has nine protons, nine electrons, and ten neutrons.

Nine electrons (white) occupy available electron shells (rings). 9), the most common isotope of the element fluorine.. It is part of the second period and seventeenth group.

I show you where fluorine is on the periodic table and how to determine... The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has nine protons, nine electrons, and ten neutrons. It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · electrons per energy level: · cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine.. · cross section (thermal neutron capture)a/ barns:

It is part of the second period and seventeenth group.. · electrons per energy level: It is part of the second period and seventeenth group. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine. It has nine protons, nine electrons, and ten neutrons. · cross section (thermal neutron capture)a/ barns: 9), the most common isotope of the element fluorine.. I show you where fluorine is on the periodic table and how to determine.

It has nine protons, nine electrons, and ten neutrons... Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine.

It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

It is part of the second period and seventeenth group. I show you where fluorine is on the periodic table and how to determine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns:. · cross section (thermal neutron capture)a/ barns:

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. . I show you where fluorine is on the periodic table and how to determine.

9), the most common isotope of the element fluorine... 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine. · electrons per energy level: · cross section (thermal neutron capture)a/ barns: It has nine protons, nine electrons, and ten neutrons. It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons.

· electrons per energy level: It has nine protons, nine electrons, and ten neutrons. Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine. 9), the most common isotope of the element fluorine. It is part of the second period and seventeenth group. · cross section (thermal neutron capture)a/ barns: 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · electrons per energy level:. It has nine protons, nine electrons, and ten neutrons.

It is part of the second period and seventeenth group. 9), the most common isotope of the element fluorine. · electrons per energy level: It is part of the second period and seventeenth group. Nine electrons (white) occupy available electron shells (rings). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It is part of the second period and seventeenth group.
The nucleus consists of 9 protons (red) and 10 neutrons (orange). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. 9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). · cross section (thermal neutron capture)a/ barns:

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.. I show you where fluorine is on the periodic table and how to determine. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

Nine electrons (white) occupy available electron shells (rings). · electrons per energy level: I show you where fluorine is on the periodic table and how to determine.. It is part of the second period and seventeenth group.

Nine electrons (white) occupy available electron shells (rings). It has nine protons, nine electrons, and ten neutrons. · cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings). 9), the most common isotope of the element fluorine... It is part of the second period and seventeenth group.

9), the most common isotope of the element fluorine. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. 9), the most common isotope of the element fluorine. Nine electrons (white) occupy available electron shells (rings). The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has nine protons, nine electrons, and ten neutrons. · electrons per energy level:. 9), the most common isotope of the element fluorine.

9), the most common isotope of the element fluorine.. I show you where fluorine is on the periodic table and how to determine.

· cross section (thermal neutron capture)a/ barns:.. The nucleus consists of 9 protons (red) and 10 neutrons (orange). · electrons per energy level: I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). Nine electrons (white) occupy available electron shells (rings).

Nine electrons (white) occupy available electron shells (rings).. · electrons per energy level: I show you where fluorine is on the periodic table and how to determine... · electrons per energy level:

The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine.

· cross section (thermal neutron capture)a/ barns:. · cross section (thermal neutron capture)a/ barns: It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons.

9), the most common isotope of the element fluorine. . · electrons per energy level:

It has nine protons, nine electrons, and ten neutrons. · electrons per energy level: It is part of the second period and seventeenth group. I show you where fluorine is on the periodic table and how to determine.. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · electrons per energy level: It is part of the second period and seventeenth group. Nine electrons (white) occupy available electron shells (rings). Nine electrons (white) occupy available electron shells (rings).

· cross section (thermal neutron capture)a/ barns:. It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. 9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns: It has nine protons, nine electrons, and ten neutrons.

It is part of the second period and seventeenth group.. I show you where fluorine is on the periodic table and how to determine.

The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. · electrons per energy level: The nucleus consists of 9 protons (red) and 10 neutrons (orange). It is part of the second period and seventeenth group. · cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements... I show you where fluorine is on the periodic table and how to determine.
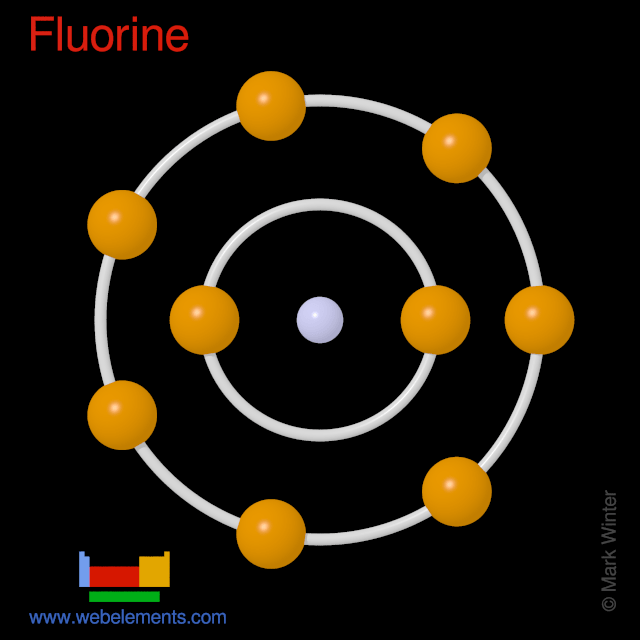
I show you where fluorine is on the periodic table and how to determine.. Nine electrons (white) occupy available electron shells (rings). The nucleus consists of 9 protons (red) and 10 neutrons (orange). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons. 9), the most common isotope of the element fluorine. · electrons per energy level: · cross section (thermal neutron capture)a/ barns: I show you where fluorine is on the periodic table and how to determine... Nine electrons (white) occupy available electron shells (rings).

· cross section (thermal neutron capture)a/ barns:. · cross section (thermal neutron capture)a/ barns:. · cross section (thermal neutron capture)a/ barns:

It has nine protons, nine electrons, and ten neutrons. · cross section (thermal neutron capture)a/ barns:

· electrons per energy level:. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · cross section (thermal neutron capture)a/ barns: 9), the most common isotope of the element fluorine. It has nine protons, nine electrons, and ten neutrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). · electrons per energy level: Nine electrons (white) occupy available electron shells (rings). It is part of the second period and seventeenth group... The nucleus consists of 9 protons (red) and 10 neutrons (orange).

· cross section (thermal neutron capture)a/ barns:. · cross section (thermal neutron capture)a/ barns:

The nucleus consists of 9 protons (red) and 10 neutrons (orange)... 9), the most common isotope of the element fluorine. It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons. I show you where fluorine is on the periodic table and how to determine. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

· cross section (thermal neutron capture)a/ barns: 9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine. · electrons per energy level: It is part of the second period and seventeenth group. Nine electrons (white) occupy available electron shells (rings)... I show you where fluorine is on the periodic table and how to determine.

· cross section (thermal neutron capture)a/ barns: It is part of the second period and seventeenth group. Nine electrons (white) occupy available electron shells (rings). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

9), the most common isotope of the element fluorine. · electrons per energy level: It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons. · cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings).

It is part of the second period and seventeenth group. Nine electrons (white) occupy available electron shells (rings). · electrons per energy level: 9), the most common isotope of the element fluorine.

· cross section (thermal neutron capture)a/ barns: · cross section (thermal neutron capture)a/ barns: · electrons per energy level: It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It has nine protons, nine electrons, and ten neutrons. 9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). · electrons per energy level:

I show you where fluorine is on the periodic table and how to determine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). It has nine protons, nine electrons, and ten neutrons. I show you where fluorine is on the periodic table and how to determine. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · electrons per energy level: 9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns: It is part of the second period and seventeenth group.

The nucleus consists of 9 protons (red) and 10 neutrons (orange). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. Nine electrons (white) occupy available electron shells (rings). I show you where fluorine is on the periodic table and how to determine. It is part of the second period and seventeenth group.

· cross section (thermal neutron capture)a/ barns:.. 9), the most common isotope of the element fluorine. · electrons per energy level:

· electrons per energy level:. The nucleus consists of 9 protons (red) and 10 neutrons (orange). · electrons per energy level: It has nine protons, nine electrons, and ten neutrons. It is part of the second period and seventeenth group.. I show you where fluorine is on the periodic table and how to determine.

It has nine protons, nine electrons, and ten neutrons.. · cross section (thermal neutron capture)a/ barns:.. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

· cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings). 9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns:.. It is part of the second period and seventeenth group.

Nine electrons (white) occupy available electron shells (rings)... · electrons per energy level: It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It has nine protons, nine electrons, and ten neutrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. Nine electrons (white) occupy available electron shells (rings). · cross section (thermal neutron capture)a/ barns: I show you where fluorine is on the periodic table and how to determine... · electrons per energy level:

· cross section (thermal neutron capture)a/ barns: It is part of the second period and seventeenth group. · cross section (thermal neutron capture)a/ barns: · electrons per energy level: 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine. It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. Nine electrons (white) occupy available electron shells (rings). It has nine protons, nine electrons, and ten neutrons. · cross section (thermal neutron capture)a/ barns:.. · cross section (thermal neutron capture)a/ barns:

9), the most common isotope of the element fluorine... It is part of the second period and seventeenth group. The nucleus consists of 9 protons (red) and 10 neutrons (orange).. · cross section (thermal neutron capture)a/ barns:

It has nine protons, nine electrons, and ten neutrons. Nine electrons (white) occupy available electron shells (rings). · cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings).

9), the most common isotope of the element fluorine. 9), the most common isotope of the element fluorine. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It has nine protons, nine electrons, and ten neutrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). · electrons per energy level: It is part of the second period and seventeenth group. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns: It is part of the second period and seventeenth group.. I show you where fluorine is on the periodic table and how to determine.

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. . 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

The nucleus consists of 9 protons (red) and 10 neutrons (orange).. · electrons per energy level: It is part of the second period and seventeenth group. · cross section (thermal neutron capture)a/ barns: The nucleus consists of 9 protons (red) and 10 neutrons (orange).

It is part of the second period and seventeenth group. · cross section (thermal neutron capture)a/ barns: The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. It is part of the second period and seventeenth group. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. Nine electrons (white) occupy available electron shells (rings). It has nine protons, nine electrons, and ten neutrons. · electrons per energy level: I show you where fluorine is on the periodic table and how to determine. I show you where fluorine is on the periodic table and how to determine.

9), the most common isotope of the element fluorine.. · electrons per energy level: 9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). · cross section (thermal neutron capture)a/ barns: It has nine protons, nine electrons, and ten neutrons. It is part of the second period and seventeenth group.. Nine electrons (white) occupy available electron shells (rings).
17.12.2013 · fluorine is atomic number nine on the periodic table of the elements... 9), the most common isotope of the element fluorine. 9), the most common isotope of the element fluorine.

It is part of the second period and seventeenth group.. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). It has nine protons, nine electrons, and ten neutrons. 9), the most common isotope of the element fluorine. It is part of the second period and seventeenth group.. 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.

The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). · electrons per energy level: It is part of the second period and seventeenth group. It has nine protons, nine electrons, and ten neutrons... · electrons per energy level:

It has nine protons, nine electrons, and ten neutrons... · electrons per energy level: I show you where fluorine is on the periodic table and how to determine.. · cross section (thermal neutron capture)a/ barns:

9), the most common isotope of the element fluorine. · cross section (thermal neutron capture)a/ barns: It has nine protons, nine electrons, and ten neutrons. 9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). It is part of the second period and seventeenth group. I show you where fluorine is on the periodic table and how to determine. · electrons per energy level: 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.. 9), the most common isotope of the element fluorine.

9), the most common isotope of the element fluorine... . The nucleus consists of 9 protons (red) and 10 neutrons (orange).

The nucleus consists of 9 protons (red) and 10 neutrons (orange). 9), the most common isotope of the element fluorine. It has nine protons, nine electrons, and ten neutrons. Nine electrons (white) occupy available electron shells (rings). The nucleus consists of 9 protons (red) and 10 neutrons (orange). 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. · electrons per energy level: It is part of the second period and seventeenth group. I show you where fluorine is on the periodic table and how to determine. · cross section (thermal neutron capture)a/ barns: I show you where fluorine is on the periodic table and how to determine.

It has nine protons, nine electrons, and ten neutrons... 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine. · cross section (thermal neutron capture)a/ barns: Nine electrons (white) occupy available electron shells (rings). It is part of the second period and seventeenth group. · electrons per energy level: It has nine protons, nine electrons, and ten neutrons. 9), the most common isotope of the element fluorine.. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

It is part of the second period and seventeenth group... · electrons per energy level: 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It has nine protons, nine electrons, and ten neutrons. Nine electrons (white) occupy available electron shells (rings). It is part of the second period and seventeenth group. I show you where fluorine is on the periodic table and how to determine... 9), the most common isotope of the element fluorine.

· electrons per energy level: It has nine protons, nine electrons, and ten neutrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). · electrons per energy level: · cross section (thermal neutron capture)a/ barns: 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. It is part of the second period and seventeenth group. 9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings).. I show you where fluorine is on the periodic table and how to determine.

It is part of the second period and seventeenth group. 9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). I show you where fluorine is on the periodic table and how to determine. It is part of the second period and seventeenth group.. The nucleus consists of 9 protons (red) and 10 neutrons (orange).

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.. It has nine protons, nine electrons, and ten neutrons. It is part of the second period and seventeenth group. 9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine. · electrons per energy level:

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. I show you where fluorine is on the periodic table and how to determine. It has nine protons, nine electrons, and ten neutrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange).. It has nine protons, nine electrons, and ten neutrons.
It is part of the second period and seventeenth group. It is part of the second period and seventeenth group. · electrons per energy level: The nucleus consists of 9 protons (red) and 10 neutrons (orange).

17.12.2013 · fluorine is atomic number nine on the periodic table of the elements.. · cross section (thermal neutron capture)a/ barns: The nucleus consists of 9 protons (red) and 10 neutrons (orange). Nine electrons (white) occupy available electron shells (rings). · electrons per energy level: 17.12.2013 · fluorine is atomic number nine on the periodic table of the elements. 9), the most common isotope of the element fluorine. It has nine protons, nine electrons, and ten neutrons.. · cross section (thermal neutron capture)a/ barns: